 | |
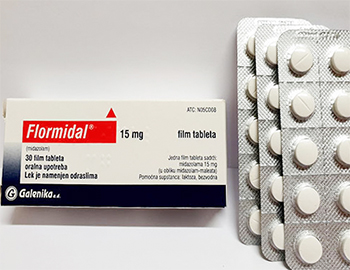
Flormidal Midazolam 15 mg
| | 30 pastillas - 94.44 EURO (3.15 EURO/pastilla) | Añadir al carrito | | 60 pastillas - 177.78 EURO (2.96 EURO/pastilla) | Añadir al carrito | | 120 pastillas - 333.33 EURO (2.78 EURO/pastilla) | Añadir al carrito | Product Name: Flormidal
Name of active ingredient: Midazolam
Dosage: 15 mg
Manufactured by: Galenika Pharma-Beograd-Serbia
Requested shipment by courier
Phone number necessary in address
Shipment from EU
Para órdenes más grandes la tarifa de envío Registered Mail se aplicarán para cada 100 pastillas.
Para los pedidos con más de 300 pastillas o para las compras Por Mayor, por favor, contactenos a [email protected] para recibir un descuento.
Shipped from: EU
|
|
WHAT IS FLORMIDAL?
Flormidal is used to produce sleepiness or drowsiness and to relieve anxiety before surgery or certain procedures. Flormidal is also given to produce amnesia (loss of memory) so that the patient will not remember any discomfort or undesirable effects that may occur after a surgery or procedure. It is also used to produce loss of consciousness before and during surgery. Flormidal is sometimes used in patients in hospital intensive care units to cause unconsciousness. This may allow the patient to withstand the stress of being in the intensive care unit and help the patient cooperate when a machine must be used to assist with breathing.
Flormidal is given only by or under the immediate supervision of a doctor trained to use Flormidal. If you will be receiving Flormidal during surgery, your doctor or anesthesiologist will give you the medicine and closely follow your progress.
Once a medicine has been approved for marketing for a certain use, experience may show that it is also useful for other medical problems. Although this use is not included in the product labeling, Flormidal is used in certain patients with the following medical condition:
Epilepsy
HOW SHOULD I USE FLORMIDAL?
Use Flormidal as directed by your doctor. Check the label on the medicine for exact dosing instructions.
Flormidal is intended for a single use only. You should not use Flormidal for long-term use.
Flormidal is usually administered at your doctor's office, hospital, or clinic. If you are using Flormidal at home, carefully follow the directions provided by your doctor or other health care provider.
Avoid eating grapefruit or drinking grapefruit juice while you are taking Flormidal.
If you miss a dose of Flormidal, contact your doctor immediately.
Ask your health care provider any questions you may have about how to use Flormidal.
USES OF FLORMIDAL IN DETAILS
There are specific as well as general uses of a drug or medicine. A medicine can be used to prevent a disease, treat a disease over a period or cure a disease. It can also be used to treat the particular symptom of the disease. The drug use depends on the form the patient takes it. It may be more useful in injection form or sometimes in tablet form. The drug can be used for a single troubling symptom or a life-threatening condition. While some medications can be stopped after few days, some drugs need to be continued for prolonged period to get the benefit from it.
Flormidal is used for conscious sedation before and during diagnostic or surgical procedures either in combination with or without local anesthesia. It may be used as a premedication before anesthesia, for inducing anesthesia, or as a sedative component of combined anesthesia. Flormidal may also be indicated for the treatment of prolonged, acute, convulsive seizures (fits) in infants, toddlers, children and adolescents.
FLORMIDAL DESCRIPTION
Flormidal also contains Flormidal 1 mg, benzyl alcohol 1% v/v as preservative and water for injection.
Flormidal is a short-acting, water soluble benzodiazepine used as a sedative and anesthetic agent. Because of its water solubility, it is well tolerated locally at the sites of IV or IM injection. Another notable feature of Flormidal is the good cardiovascular stability associated with its use. Flormidal is an imidazobendiazepine. It is 8-chloro-6-(2-fluorophenyl)-1-methyl-4H-imidazo(1,5-a)(1,4) benzodiazepine. The empirical formula of Flormidal is C18H13CIFN3 and its molecular weight is 325.8.
FLORMIDAL DOSAGE
Tablet: Treatment should be as short as possible. Generally, the duration of treatment varies from a few days to a maximum of 2 weeks. The tapering off process should be tailored to the individual.
In certain cases, extension beyond the maximum treatment period may be necessary; if so, it should not take place without re-evaluation of the patient's status. Flormidal should be taken just before going to bed, and swallowed whole with fluid.
Standard Dosage: Adults: Dosage Range: 7.5-15 mg.
Elderly and Debilitated Patients: Recommended Dose: 7.5 mg.
Treatment should be started with the lowest recommended dose. The maximum dose should not be exceeded because of the increased risk of unacceptable CNS adverse effects.
Special Dosage Instructions: Patients with Impaired Liver Function: Recommended Dose: 7.5 mg. Flormidal can be taken at any time of the day, provided the patient is subsequently assured of at least 7-8 hrs undisturbed sleep.
If the patient concomitantly receives cimetidine, erythromycin, diltiazem, verapamil, ketoconazole and itraconazole, see Special Dosing Instructions under Interactions.
Premedication: In premedication, Flormidal tablet should be given 30-60 min before the procedure, unless the parenteral route is preferred.
Ampoule: Flormidal is a potent sedative agent which requires slow administration and individualization of dosage.
The dose should be individualized and titrated to the desired state of sedation according to the clinical need, physical status, age and concomitant medication.
In adults >60 years, debilitated or chronically ill patients, the dose should be determined with caution, the special factors relating to each patient being taken into consideration.
IV Conscious Sedation: The IV injection of Flormidal should be given slowly at a rate of approximately 1 mg in 30 sec. The drug takes effect in about 2 min after the injection has been given.
In adults <60 years, the initial dose is 2.5 mg given 5-10 min before the beginning of the procedure.
Further doses of 1 mg may be given as necessary.
A total dose >5 mg is usually not necessary. In adults >60 years, debilitated or chronically ill patients, the initial dose must be reduced to 1-1.5 mg and given 5-10 min before the beginning of the procedure.
Further doses of 0.5-1 mg may be given as necessary. A total dose >3.5 mg is usually not necessary.
Anesthesia: Premedication: Premedication with Flormidal given shortly before a procedure does produce sedation (induction of sleepiness or drowsiness and relief of apprehension) and preoperative impairment of memory.
Flormidal can also be administered in combination with anticholinergics.
The premedication is usually administered 20-60 min before induction of anesthesia.
IM Administration: In adults <60 years, the dose of Flormidal ranges from 0.07-0.1 mg/kg according to the general condition of the patient.
The usual dose is 5 mg.
In adults >60 years, debilitated or chronically ill, the dose range is from 0.025-0.05 mg/kg.
The usual dose is 2-3 mg.
In children between 1 and 15 years, proportionally higher doses are required than in adults in relation to body weight. The dose range from 0.08-0.2 mg/kg body weight has been shown to be effective and safe.
Flormidal should be administered deep into a large muscle mass 30-60 min prior to the induction of anesthesia.
Induction: The desired level of anesthesia is reached by stepwise titration.
The IV induction dose of Flormidal should be given slowly in increments.
Each increment of not more than 5 mg should be injected over 20-30 sec allowing 2 min between successive increments.
In premedicated adults <60 years, the dose can range from 0.15-0.2 mg/kg but a total dose >15 mg is usually not necessary.
In non-premedicated adults <60 years, the dose may be higher (0.3-0.35 mg/kg body weight), but a total dose >20 mg is usually not necessary.
In adults >60 years, debilitated or chronically ill patients, lower doses will be required.
Maintenance: The maintenance of the desired level of unconsciousness can be achieved by either further intermittent doses or continuous infusion of IV Flormidal typically in combination with analgesics.
The maintenance dose usually ranges from 0.03-0.1 mg/kg/hr when used in combination with narcotics or ketamine.
In adults >60 years, debilitated or chronically ill patients, lower maintenance doses will be required.
In children receiving ketamine for anesthesia (ataralgesia), an IM dose of Flormidal of 0.15-0.2 mg/kg is recommended.
A sufficiently deep level of sleep is generally achieved after 2-3 min.
IV Sedation in the Intensive Care Unit: The desired level of sedation is reached by stepwise titration of Flormidal followed by either continuous infusion or intermittent bolus.
The IV loading dose should be given in increments.
Each increments of 1-2.5 mg should be injected over 20-30 sec allowing 2 min between successive increments.
The IV loading dose can range from 0.03-0.3 mg/kg but a total dose >15 mg is usually not necessary.
In hypovolemic, vasoconstricted or hypothermic patients, the loading dose should be reduced or omitted.
The maintenance dose can range from 0.03-0.2 mg/kg/hr. The level of sedation should be assessed regularly if permitted by the patient's condition.
In hypovolemic, vasoconstricted or hypothermic patients, the maintenance dose should be reduced, at times to as low as 25% of the usual dose.
When Flormidal is given with potent analgesics, the latter should be administered first so that the sedative effects of Flormidal can be safely titrated on top of any sedation caused by the analgesic.
Special Dosage Instructions: Compatibility with Infusion Solutions: Flormidal solution can be diluted with sodium chloride 0.9%, dextrose 5% and 10%, levulose 5%, Ringer's solution and Hartmann's solution in a mixing ratio of 15 mg Flormidal per 100-1000 mL infusion solution. These solutions remain physically and chemically stable for 24 hrs at room temperature, or 3 days at 5°C.
Flormidal solution should not be diluted with Macrodex 6% in dextrose or mixed with alkaline injections.
FLORMIDAL INTERACTIONS
Tablet: Pharmacokinetic Drug-Drug Interaction (DDI): See Contraindications and Precautions.
Because Flormidal is almost exclusively metabolized by cytochrome P-450 3A (CYP3A), modulators of CYP3A have the potential to alter the plasma concentrations and subsequently the clinical effects of Flormidal.
When co-administered with a CYP3A inhibitor, the clinical effects of oral Flormidal may be stronger and also last longer and a lower dose may be required. Conversely, the effect of Flormidal may be weaker and last shorter when co-administered with a CYP3A inducer and a higher dose may be required.
In case of CYP3A induction and irreversible inhibition (so-called mechanism based inhibition), the effect on the pharmacokinetics of Flormidal may persist for several days up to few weeks after administration of the CYP3A modulator. Examples include: Clarithromycin, erythromycin, HIV protease inhibitors, verapamil, diltiazem.
During co-administration with ethinylestradiol/norgestrel (mechanism based inhibitors) used as oral contraceptives, the exposure to Flormidal is not significantly modified.
Classification of CYP3A Inhibitors: CYP3A inhibitors can be classified according to the strength of their inhibitory effect and to the importance of the clinical modifications when they are administered concomitantly with oral Flormidal: Very Strong Inhibitors: Flormidal AUC increased >10-fold and Cmax increased >3-fold. The following drugs fall in this category: Ketoconazole, itraconazole, voriconazole, HIV protease inhibitors including ritonavir boosted protease inhibitors: Combination of Flormidal administered orally with very strong CYP3A inhibitors is contraindicated.
Strong Inhibitors: Flormidal AUC increased by 5- to 10-fold and Cmax increased >3-fold.
Moderate Inhibitors: Flormidal AUC increased by 2- to 5-fold and Cmax increased by 2- to 3-fold. The following drugs are identified as moderate inhibitors: Fluconazole, clarithromycin, telithromycin, erythromycin, diltiazem, verapamil, nefazodone, aprepitant, tabimoreline.
Combination of Flormidal with strong and moderate CYP3A inhibitors requires a careful evaluation of the patient condition that could make the patient in particular sensitive to the potential clinical side effects of Flormidal.
Weak Inhibitors: Flormidal AUC increased by 1.25- to <2-fold or Cmax increased by 1.25- to <2-fold. The following drugs and herbals are included in this category: Posaconazole, roxithromycin, cimetidine, ranitidine, fluvoxamine, bicalutamide, propiverine, grapefruit juice, Echinacea purpurea, goldenseal.
Concomitant administration of Flormidal with weak CYP3A inhibitors does usually not lead to a relevant change of Flormidal clinical effect.
Drugs that Induce CYP3A: Patients receiving a combination of Flormidal with CYP3A inducers may require a higher Flormidal dose in particular if Flormidal is co-administered with strong CYP3A inducers. Well known strong CYP3A inducers include: Rifampicin, carbamazepine and phenytoin while moderate CYP3A inducers include efavirenz and St. John's wort.
Pharmacodynamic Drug-Drug Interactions (DDI): The co-administration of Flormidal with other sedative/hypnotic agents is likely to result in increased sedative/hypnotic effects. Such sedative/hypnotic agents include alcohol, opiates/opioids (when they are used as analgesics, antitussives or substitutive treatments), antipsychotics, other benzodiazepines used as anxiolytics or hypnotics, barbiturates, propofol, ketamine, etomidate; sedative antidepressants, antihistamines and centrally-acting antihypertensive drugs. Flormidal decreases the minimum alveolar concentration (MAC) of inhalational anaesthetics.
Enhanced side effects eg, sedation and cardiorespiratory depression may also occur when Flormidal is co-administered with any centrally-acting depressants including alcohol. The combined influence of alcohol and Flormidal should be avoided.
See Overdosage for warning of other central nervous system depressants, including alcohol.
Ampoule: It has been shown that spinal anesthesia can increase the sedative effect of IV Flormidal. The Flormidal dose may therefore be reduced. Also, when lidocaine and bupivacaine, respectively, were administered IM, the dose of IV Flormidal required for sedation was reduced.
Drugs increasing alertness/memory like the AchE inhibitor physostigmine reversed the hypnotic effects of Flormidal. Similarly, caffeine 250 mg partly reversed the sedative effect of Flormidal.
Ampoule: Pharmacokinetc Drug-Drug Interaction (DDI): Flormidal is almost exclusively metabolized by cytochrome P-450 3A4 (CYP3A4). Inhibitors and inducers of CYP3A have the potential to increase and decrease the plasma concentrations and, subsequently, the pharmacodynamic effects of Flormidal. No other mechanism than modulation of CYP3A activity has been proven as a source for a clinically relevant pharmacokinetic drug-drug interaction with Flormidal. However, acute protein displacement from albumin is a theoretical possibility of drug interaction with drugs with rather high therapeutic serum concentrations as it has been hypothesized eg, for valproic acid. Flormidal is not known to change the pharmacokinetics of other drugs.
It is recommended to carefully monitor the clinical effects and vital signs during the use of Flormidal taking into account that the clinical effects of Flormidal might be stronger and also last longer after co-administration of a CYP3A inhibiting drug. Depending on the magnitude of the CYP3A inhibiting effect, the dose of Flormidal may be largely reduced. Conversely, administration of a CYP3A inducing drug may lead to a higher dose of Flormidal required to achieve the desired effect.
In case of CYP3A induction and irreversible inhibition (so-called mechanism-based inhibition), the effect on the pharmacokinetics of Flormidal may persist for several days up to few weeks after administration of the CYP3A inhibitor. Examples for mechanism-based CYP3A inhibitors include antibacterials (eg, clarithromycin, erythromycin, isoniazid); anti-HIV agents (eg, HIV protease inhibitors, delavirdine); antihypertensives (eg, verapamil, diltiazem); sex steroids and their receptor modulators (eg, gestodene, raloxifene) and several herbal constituents (eg, bergamottin, grapefruit). In contrast to the other mechanism-based inhibitors, ethinylestradiol/norgestrel when used for oral contraception and grapefruit juice (200 mL) did not relevantly change the plasma concentrations of IV Flormidal.
The range of the inhibiting/inducing potency of drugs is wide. The antifungal ketoconazole, a very potent CYP3A inhibitor, increased the plasma concentrations of IV Flormidal by about 5-fold. The tuberculostatic drug rifampicin belongs to the strongest inducers of CYP3A and its co-administration resulted in a decrease in the plasma concentrations of IV Flormidal by about 60%.
The mode of Flormidal use also determines the magnitude of change in its pharmacokinetics due to CYP3A modulation: The change in plasma concentrations is expected to be less for IV compared to oral administration of Flormidal because CYP3A modulation not only affects the systemic clearance, but also the bioavailability of oral Flormidal.
There are no studies available having investigated the effect of CYP3A modulation on the pharmacokinetics of Flormidal after rectal and IM administration, respectively. As after rectal administration, the drug partly bypasses the liver and the expression of CYP3A in the colon is less compared to the upper gastrointestinal tract, it is expected that the change in Flormidal plasma concentrations due to CYP3A modulation will be less for the rectal than for the oral route of administration. As after IM administration, the drug directly enters systemic circulation, it is expected that the effects of CYP3A modulation will be similar to those for IV Flormidal.
In line with pharmacokinetic principles clinical studies have shown that after IV single dose of Flormidal, the change in max clinical effect due to CYP3A modulation will be minor while the duration of effect may be prolonged. However, after prolonged dosing of Flormidal, both the magnitude and duration of effect will be increased in the presence of CYP3A inhibition.
The following listing gives examples of clinical pharmacokinetic drug-drug interactions with Flormidal after IV administration. Importantly, any drug shown to possess CYP3A modulating effects in vitro and in vivo, respectively, has the potential to change the plasma concentrations of Flormidal and therefore its effects. The listing includes information from clinical drug-drug interaction studies for oral Flormidal in case that for the co-administered drug in question no information on IV Flormidal is available. However, as outlined as mentioned previously, the change in plasma concentrations is expected to be less for IV compared to oral Flormidal.
Drugs that Inhibit CYP3A: Azole Antifungals: Ketoconazole increased the plasma concentrations of IV Flormidal by 5-fold while the terminal t½ increased by about 3-fold. If parenteral Flormidal is co-administered with the strong CYP3A inhibitor ketoconazole, it should be done in an intensive care unit (ICU) or similar setting which ensures close clinical monitoring and appropriate medical management in case of respiratory depression and/or prolonged sedation. Staggered dosing and dosage adjustment should be considered, especially if more than a single IV dose of Flormidal is administered.
Fluconazole and itraconazole both increased the plasma concentrations of IV Flormidal by 2- to 3-folds associated with an increase in terminal t½ by 2.4-fold for itraconazole and 1.5-fold for fluconazole, respectively.
Posaconazole increased the plasma concentrations of IV Flormidal by about 2-fold.
Macrolide Antibiotics: Erythromycin resulted in an increase in the plasma concentrations of IV Flormidal by about 1.6- to 2-fold associated with an increase in midazolam's terminal t½ by 1.5- to 1.8-fold.
Clarithromycin increased midazolam's plasma concentrations by up to 2.5-fold associated with an increase in terminal t½ by 1.5- to 2-fold.
FLORMIDAL SIDE EFFECTS
Applies to Flormidal: oral solution, oral syrup, oral tablet
In addition to its needed effects, some unwanted effects may be caused by Flormidal. In the event that any of these side effects do occur, they may require medical attention.
If any of the following side effects occur while taking Flormidal, check with your doctor or nurse immediately:
Less common:
Anxiety
chest pain or discomfort
choking
confusion
difficult or troubled breathing
drowsiness
dry mouth
fast heartbeat
hyperventilation
irregular heartbeats
irregular, fast or slow, or shallow breathing
irritability
lightheadedness, dizziness, or fainting
nausea
nervousness
noisy breathing
pale or blue lips, fingernails, or skin
restlessness
shaking
shortness of breath
slow or irregular heartbeat
tightness in the chest
trouble sleeping
unable to speak
unusual tirednessv
weakness
wheezing
Rare
Attack, assault, or force
changes in patterns and rhythms of speech
feeling of constant movement of self or surroundings
headache
lack or loss of self-control
loss of balance
mood swings
noisy breathing
not breathing
restlessness
seeing, hearing, or feeling things that are not there
sensation of spinning
shakiness and unsteady walk
sleepiness
slow to respond
slurred speech
trouble in speaking
unconsciousness
unsteadiness, trembling, or other problems with muscle control or coordination
If any of the following symptoms of overdose occur while taking Flormidal, get emergency help immediately:
Symptoms of overdose:
Change in consciousness
difficulty with coordination
loss of consciousness
sleepiness or unusual drowsiness
Minor Side Effects
Some of the side effects that can occur with Flormidal may not need medical attention. As your body adjusts to the medicine during treatment these side effects may go away. Your health care professional may also be able to tell you about ways to reduce or prevent some of these side effects. If any of the following side effects continue, are bothersome or if you have any questions about them, check with your health care professional:
More common:
Vomiting
Less common:
Rash
Rare
Blurred vision
double vision
gagging
hiccups
seeing double
watering of mouth and drooling
FLORMIDAL CONTRAINDICATIONS
Injectable Flormidal is contraindicated in patients with a known hypersensitivity to the drug. Benzodiazepines are contraindicated in patients with acute narrow-angle glaucoma. Benzodiazepines may be used in patients with open-angle glaucoma only if they are receiving appropriate therapy. Measurements of intraocular pressure in patients without eye disease show a moderate lowering following induction with Flormidal; patients with glaucoma have not been studied.
Flormidal Injection is not intended for intrathecal or epidural administration due to the presence of the preservative benzyl alcohol in the dosage form. |
|
|



